Device Description
The B-Clamp® is a laparoscopically implanted, silicone covered titanium clamp with a computer designed low closing pressure placed vertically around the stomach parallel to the lesser curvature of the stomach, which would separate the stomach into a restricted medial segment where food would pass and an excluded larger lateral gastric segment. The device restricts the amount of food consumption in severely obese patients without the need for cutting, stapling, bypassing of the stomach or the intestines, or device adjustment.
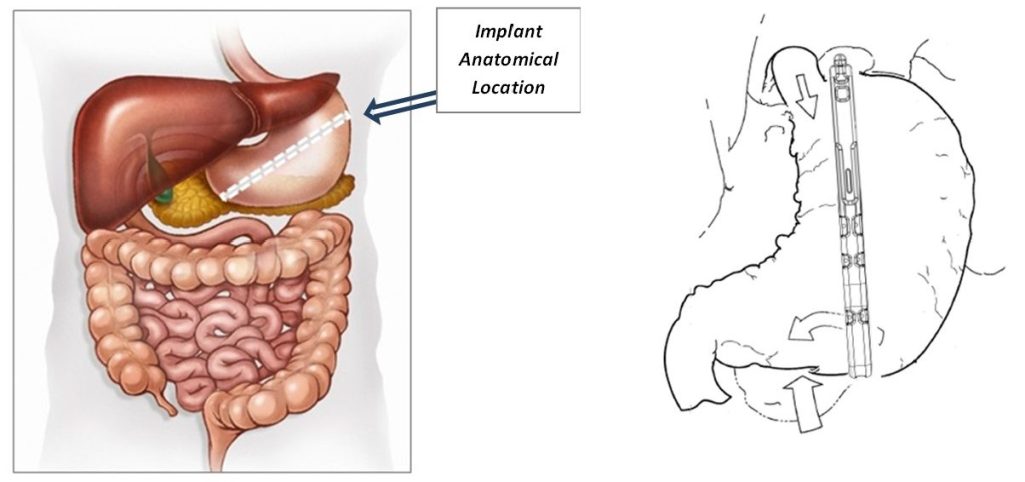
The B-Clamp® is intended for treatment of obesity patients which, in function, partitions the stomach in two vertical sections, when closed, latched and sutured in place. Commercially available surgical graspers or clamps are recommended for proper insertion, positioning, and latching of the B-Clamp® device. The lesser curvature segment is the Magenstrasse, while the greater curvature segment including the fundus is excluded from nutritional contact. The clamp at its bottom end has an aperture by which gastric juices created by the fundus and the body can empty into the atrum. Additionally, the B-Clamp® acts as a restrictive procedure, by creating a small lumen for a vertical passageway of the nutrients along the lesser curvature.
Enough pressure walls (~4gm/mm which is less than the pressure of an inflated band) is applied on the gastric with the B-Clamp® closed and sutured in place, such that the formed gastric tube is maintained closed, without causing ischemia, ulcerations or injuries to the treated region. The B-Clamp® hinge section (aperture) allows gastric contents to flow freely from the fundus and body of the stomach into the antrum and pylorus. At the time of implantation, the gastric lumen should be calibrated using a standard bougie (36 F). The B-Clamp® does not alter the anatomical and/or physiological structure of the digestive system, given that there is no removal or cutting of tissue during implantation.
The B-Clamp® has been designed and developed as a permanent (long-term) implant that is removable.
B-Clamp® Implant Anatomical Location
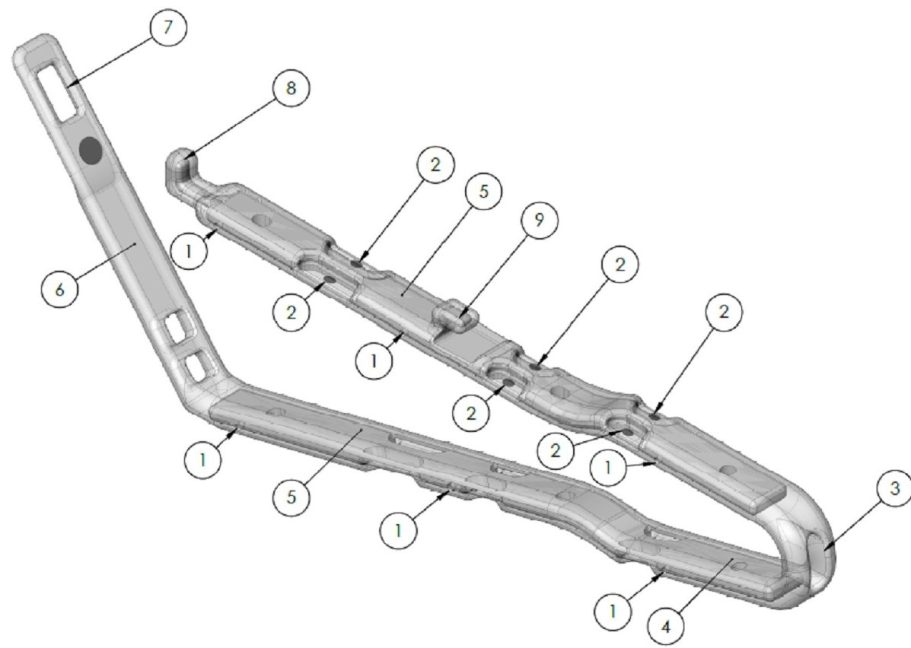
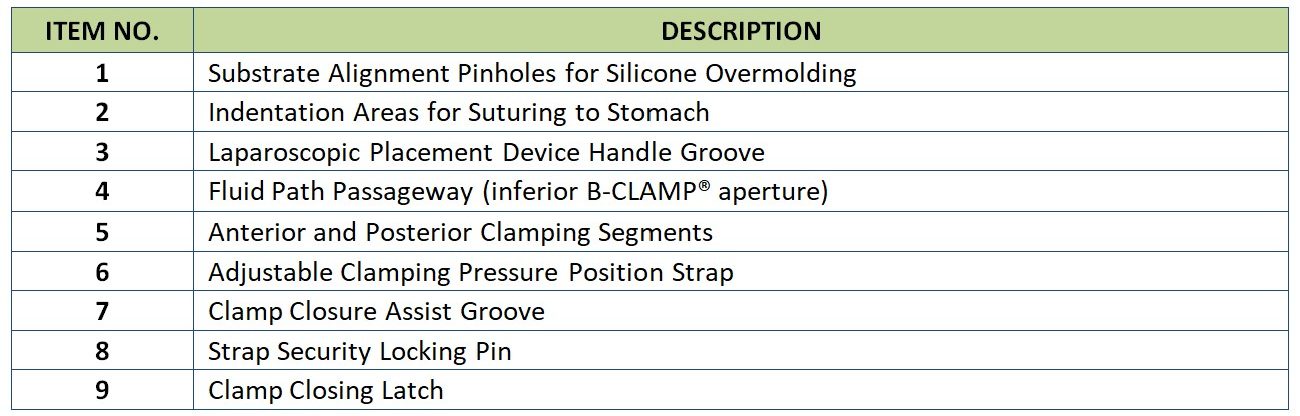
Indications for Use
The B-Clamp® is indicated for weight reduction for patients with obesity, with a Body Mass Index (BMI) of at least 35 kg/m2 or between 30 kg/m2 and 50 kg/m2 with one or more obesity related comorbid conditions. It is indicated for use only in adult patients (18 years old or older) who have previously failed in more conservative weight reduction alternatives, such as supervised diet, exercise or behavior modification programs. Patients who elect to have this surgery must make the commitment to accept significant changes in their eating habits for the rest of their lives.
Complications
Patients should be advised that the B-Clamp® is a long-term implant. Explant (removal) may be indicated at any time. Management of adverse reactions may include explantation. Revisional surgery for repositioning and replacement may also be indicated to correct displacements, and achieve patient satisfaction.
Slippage and Erosion
The most common complication is displacement or slippage, which occurs in about 1.5% of cases. Rarely, it can become an emergency when it causes gastric outlet obstruction. In most cases the B-Clamp® can be repositioned, if not , the B-Clamp® is easily removable.
With the current anchoring techniques, the displacement/slippage rate has been less than 1%.
The current erosion rate is approximately 1%. All erosions to date have been associated with prior slippage of the clip. The B-Clamp® may be removed via endoscopy or via laparoscopy.
Bleeding and Infection
To date, we know of no patient requiring transfusion, of any intra abdominal infections, of having the procedure being converted to open, or of any deaths.
© Copyright. All rights reserved.
We need your consent to load the translations
We use a third-party service to translate the website content that may collect data about your activity. Please review the details in the privacy policy and accept the service to view the translations.
